HIFU Device Ultimate Guide 2024: How to Choose the Best Non-Invasive Beauty Machine_15
The Complete Guide to HIFU Devices for Sale in 2024
The global HIFU market is projected to reach $1.2 billion by 2027, growing at 12.3% CAGR according to Grand View Research. As demand for non-invasive cosmetic procedures surges, professionals and consumers alike face challenges identifying quality HIFU devices that deliver real results without compromising safety. This comprehensive guide will walk you through essential purchasing considerations, technical specifications, industry trends, and frequently asked questions about HIFU technology.
HIFU Device Buying Guide: Professional vs Home Use
When evaluating HIFU devices for sale, consider these key factors:
| Feature | Professional HIFU | Home HIFU |
|---|---|---|
| Energy Output | 5-10MHz | 1-3MHz |
| Treatment Depth | 4.5mm-13mm | 1.5mm-3mm |
| FDA Clearance | Required | Recommended |
7 Must-Check Features for Professional HIFU Machines
- FDA 510(k) clearance documentation
- Adjustable depth penetration (3-13mm)
- Real-time temperature monitoring
- Dual-frequency capability
- CE/RoHS certification
- Minimum 1000-hour transducer lifespan
- Two-year manufacturer warranty
HIFU Technology Deep Dive
High-Intensity Focused Ultrasound works by delivering focused thermal energy to subcutaneous layers at precise depths. The diagram below illustrates how HIFU stimulates collagen production in the SMAS layer without damaging surface skin.
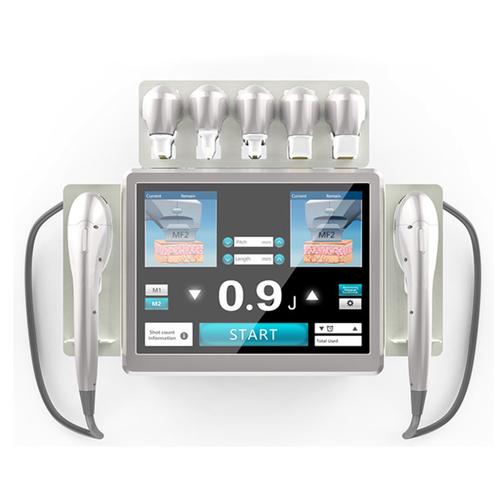
How HIFU Compares to Alternative Technologies
Unlike RF or laser treatments, HIFU devices:
- Require no downtime
- Show cumulative results over 3-6 months
- Can target deeper facial muscles
- Maintain consistent energy at all skin tones
2024 HIFU Industry Trends
The European market leads in professional HIFU adoption (38% share), while Southeast Asia shows fastest growth (18% YoY). New FDA guidelines now require:
- Enhanced safety protocols for home devices
- Clearer labeling of treatment indications
- Standardized energy output measurements
HIFU FAQ
Is HIFU safe for all skin types?
FDA-cleared devices are safe for Fitzpatrick I-VI when used as directed. Avoid treatments on:
- Active acne areas
- Metal implant locations
- Pregnant women
What certifications should I verify?
Look for FDA 510(k), CE, ISO 13485, and RoHS compliance. Professional devices require medical device registration in most countries.
How to handle customs clearance?
Most countries classify HIFU as Class II medical devices. Prepare:
- Certificate of Origin
- FDA/CE certificates
- Commercial invoice with HS code 9018.90.80


















