Face Skin Analysis Scanner Machine Ultimate Guide 2024: How to Choose the Best Skin Diagnostic Device_81
The Complete Guide to Face Skin Analysis Scanner Machines in 2024
The global skin analysis devices market is projected to reach $1.2 billion by 2027, growing at 8.3% CAGR (Statista). As consumers become increasingly skincare-conscious, professionals and clinics struggle with outdated diagnostic methods that deliver inconsistent results. This comprehensive guide will walk you through everything from technical specifications to emerging trends in facial skin analysis technology.
Face Skin Analysis Scanner Buying Guide
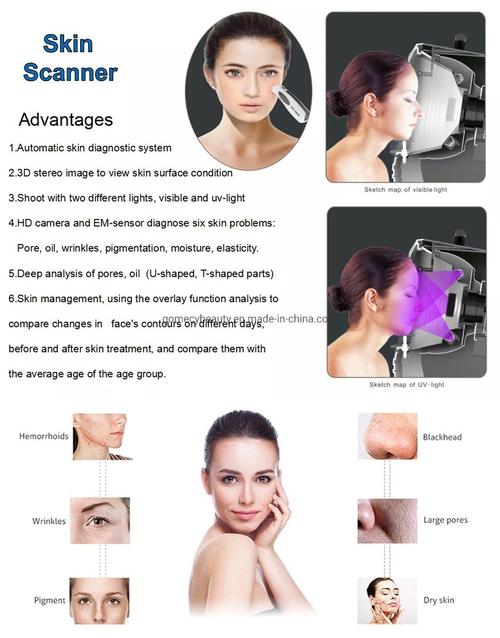
When investing in skin analysis equipment, these 7 features are non-negotiable:
- Multi-spectral imaging capability
- Minimum 8MP resolution
- AI-powered analysis software
- Cross-polarized lighting
- UV fluorescence detection
- Portable design (under 5lbs)
- Cloud-based data storage
Comparison: Professional vs Consumer-grade Scanners
| Feature | Medical-grade | Spa/Retail |
|---|---|---|
| Accuracy | ±2% | ±15% |
| Analysis Depth | Subdermal | Epidermal |
Technical Specifications Explained
Modern skin scanners utilize:
- RBX technology for pigment analysis
- 3D topographic mapping
- TEWL sensors for barrier function
Manufacturers must comply with:
- FDA Class II medical device standards
- ISO 13485 certification
- GDPR for data privacy
Industry Trends
Key developments include:
- 42% growth in AI-powered home devices (Euromonitor)
- New EU regulations on cosmetic claims verification
- Rising demand in Southeast Asian markets
FAQ
Q: Can these scanners detect skin cancer?
A: Only FDA-cleared medical devices can screen for malignancies.
Q: What certifications are required?
A: CE marking for Europe, FDA for US markets.