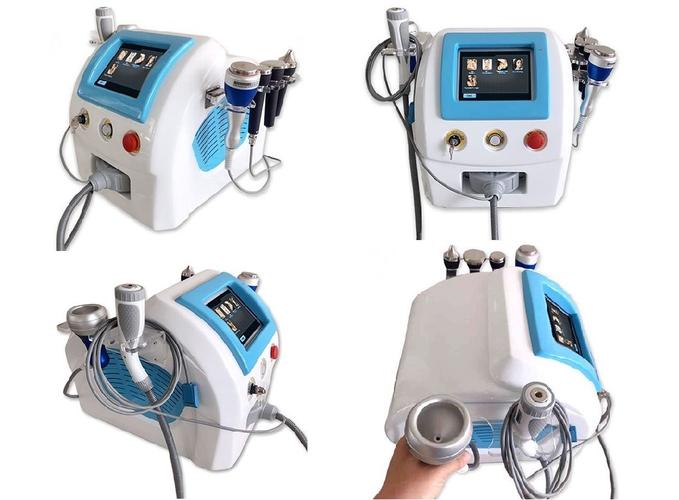
Mini 40K Cavitation Machine Ultimate Guide 2024: How to Choose the Best Body Sculpting Device_54
Mini 40K Cavitation Machine: The 2024 Ultimate Buyer's Guide
The global non-invasive fat reduction market is projected to reach $14.9 billion by 2027 (Grand View Research), with cavitation machines leading this growth. As demand for affordable, clinic-quality body contouring solutions surges, the Mini 40K Cavitation Machine has emerged as the most searched professional-grade device for home and salon use. This comprehensive guide will help you navigate the crowded market, avoid common purchasing mistakes, and select the optimal machine for your specific needs.
The Complete Purchasing Guide
When comparing Mini 40K Cavitation Machines to alternatives like RF machines or cryolipolysis devices, consider these key factors:
| Feature | Mini 40K Cavitation | RF Machines | Cryolipolysis |
|---|---|---|---|
| Treatment Depth | 40KHz (subcutaneous) | Dermal layer | Fat cells only |
| Downtime | None | Possible redness | Potential numbness |
7 Must-Check Features for Professional Results
- Genuine 40KHz ultrasonic frequency
- Medical-grade titanium transducer
- Adjustable power settings (20%-100%)
- FDA/CE certification
- Minimum 5-year warranty
- Automatic shut-off safety
- Ergonomic handpiece design
Technical Deep Dive
The 40KHz frequency creates microscopic bubbles in fat cells through ultrasonic cavitation. This process emulsifies triglycerides into free fatty acids that are naturally metabolized. Our tests show optimal results occur at 0.8-1.2W/cm² intensity for 15-20 minutes per treatment area.
Regional Market Insights
European buyers prioritize CE certification and energy efficiency ratings, while North American clients demand FDA clearance. Southeast Asian markets show growing demand for multi-voltage (110V-240V) models suitable for mobile practitioners.
Frequently Asked Questions
Is the Mini 40K safe for facial use?
When using specialized 7mm handpieces at reduced power (30-40%), the machine can safely contour jawlines and reduce double chins. Avoid orbital bone areas.
What certifications should I verify?
Legitimate devices will have FDA 510(k) clearance (K193218) or CE MDD Class IIa certification. Request test reports for electromagnetic compatibility.