EMS Body Toning Device Ultimate Guide 2024: How to Choose the Best Muscle Stimulator_50
The Complete Guide to EMS Body Toning Devices in 2024
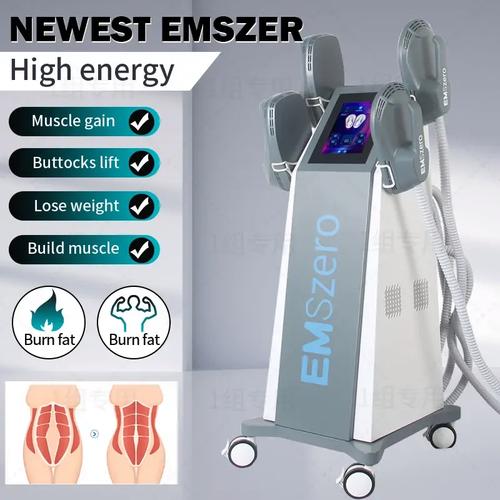
The global EMS (Electrical Muscle Stimulation) fitness equipment market is projected to reach $1.2 billion by 2027, growing at 8.3% CAGR according to Market Research Future. As more consumers seek efficient workout solutions, EMS body toning devices have emerged as revolutionary tools for muscle development and body contouring. This guide will help you navigate the complex landscape of EMS technology, from understanding core specifications to identifying genuine FDA-approved devices that deliver real results.
EMS Device Buying Guide: 7 Critical Factors
When comparing EMS body toning devices, these specifications determine effectiveness:
| Feature | Professional Grade | Consumer Grade |
|---|---|---|
| Pulse Frequency | 1-150Hz | 1-100Hz |
| Channels | 8-12 | 4-6 |
| Programs | 20+ | 5-10 |
7 Must-Check Features for Home EMS Devices
- FDA or CE medical certification
- Adjustable intensity (at least 100 levels)
- Minimum 6 electrode pads
- Lithium-ion battery with 8+ hour runtime
- Bluetooth/WiFi connectivity
- Waterproof construction (IPX6+)
- 2-year manufacturer warranty
Technical Deep Dive: How EMS Toning Works
Modern EMS devices use biphasic waveforms that mimic natural nerve signals. The optimal frequency range for muscle toning is 20-50Hz, while fat burning occurs at 7-20Hz. Leading manufacturers like Compex and PowerDot incorporate proprietary algorithms that automatically adjust pulse width (100-400μs) based on muscle response.
Regional Market Differences
European models typically emphasize medical rehabilitation features, while Asian manufacturers focus on aesthetic body shaping. North American consumers prefer hybrid devices that combine EMS with TENS functionality.
FAQ
Q: Can EMS replace traditional workouts?
A: While effective for muscle activation, EMS works best when combined with conventional exercise (30% time reduction according to German Sport University studies).
Q: What certifications matter?
A: Look for FDA Class II, CE 93/42/EEC, or ISO 13485 compliance for medical-grade devices.