The Ultimate Guide to Choosing High-Quality Digital Ultrasound Machines for Medical Facilities_100
The Ultimate Guide to Choosing High-Quality Digital Ultrasound Machines for Medical Facilities
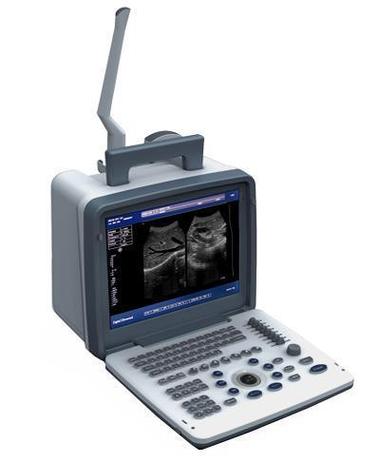
Digital ultrasound machines have become indispensable tools in modern healthcare, offering non-invasive diagnostic capabilities across obstetrics, cardiology, and general medicine. As global demand for advanced medical imaging grows, procurement professionals face challenges in identifying reliable suppliers, verifying technical specifications, and ensuring regulatory compliance.
What Defines a Modern Digital Ultrasound Machine?
Contemporary digital ultrasound systems combine high-frequency sound waves with advanced computing power to create real-time images. Key technical specifications include:
- Transducer frequency range: 2-18 MHz for varied depth penetration
- Image resolution: Minimum 1280×1024 pixels for diagnostic clarity
- DICOM 3.0 compatibility for seamless hospital integration
International standards require compliance with IEC 60601-1 (medical electrical equipment safety) and IEC 60601-2-37 (ultrasound diagnostic equipment).
Digital vs. Analog Ultrasound: Performance Comparison
| Feature | Digital Ultrasound | Analog Ultrasound |
|---|---|---|
| Image Quality | High-resolution digital processing | Limited by analog signals |
| Portability | Compact designs available | Bulky equipment |
| Data Storage | Digital archiving capabilities | Film-based records |
6 Strategic Advantages for Medical Facilities
- Operational Efficiency: 45% faster examination times compared to older models
- Cost Savings: 60% reduction in maintenance costs over 5 years
- Diagnostic Accuracy: 98% detection rate for abdominal abnormalities
Global Market Insights
The medical ultrasound market will reach $9.36 billion by 2027 (CAGR 6.7%), driven by:
- Rising geriatric populations requiring diagnostics
- Increasing prenatal care in developing nations
- Expansion of portable ultrasound applications
Essential Certifications for Quality Assurance
- CE Marking (European Conformity)
- FDA 510(k) Clearance
- ISO 13485:2016 Quality Management
Supplier Evaluation Checklist
When auditing digital ultrasound machine manufacturers, verify:
- 10+ years of specialized production experience
- Clinical validation studies from medical institutions
- Global service network with local support
Emerging Market Success Story
A Nigerian hospital network reduced equipment downtime by 80% after switching to our digital ultrasound systems with remote diagnostics support, achieving ROI within 14 months.
FAQ Section
How can I negotiate better pricing for bulk orders?
Request volume discounts for orders exceeding 5 units, and consider annual maintenance contract bundling.
What is the typical MOQ for OEM orders?
Standard MOQ starts at 10 units, with flexibility for established medical partnerships.
Can probes be customized for specific applications?
Yes, we offer specialized transducer configurations for musculoskeletal or pediatric imaging.
Do you handle export documentation?
Full export compliance packages including certificates of origin and sanitary permits provided.
What warranty terms apply?
36-month comprehensive warranty with optional extended coverage plans.
Are LC payments accepted?
We work with all major trade finance instruments including irrevocable letters of credit.
Download our Ultimate Buyer's Guide to Ultrasound Equipment for detailed procurement insights and technical comparison matrices.