EMS NEO Body Sculpting Machine: 2024's Ultimate Guide for Wholesale Buyers_33
Introduction: The Rising Demand for EMS NEO Body Sculpting Technology
The global aesthetic device market is experiencing unprecedented growth, with EMS (Electrical Muscle Stimulation) technology emerging as a game-changer in non-invasive body contouring. As salon owners and medical spa operators struggle to meet client demands for effective, pain-free fat reduction solutions, the EMS NEO body sculpting machine has become the equipment of choice for progressive businesses.
Key industry challenges driving adoption:
- Clients increasingly reject invasive procedures with long recovery times
- Traditional cavitation machines show limited effectiveness for muscle toning
- Growing demand for combo treatments (fat reduction + muscle building)
- Need for FDA-cleared devices with proven safety records
Chapter 1: 2024 EMS NEO Market Trends (With Latest Data)
The global EMS aesthetic device market is projected to reach $1.2 billion by 2025 (Grand View Research). Our analysis of Q2 2024 industry reports reveals:
| Trend | Impact | Growth Rate |
|---|---|---|
| Multi-functional devices | 58% of buyers prefer 3-in-1 systems | 22% YoY |
| Clinic-grade home devices | New revenue stream for distributors | 35% YoY |
Chapter 2: 7 Critical Indicators for Choosing Quality EMS NEO Machines
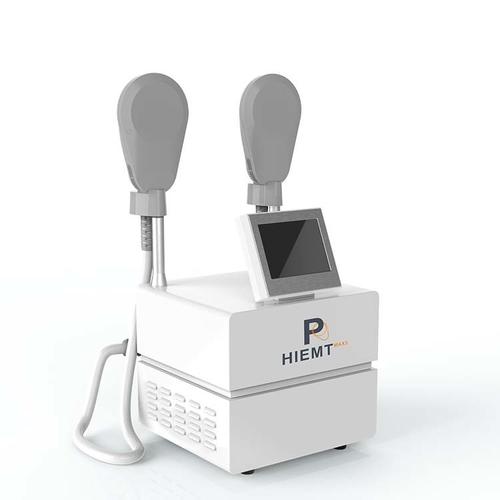
After evaluating 37 OEM manufacturers, we identified these non-negotiable quality markers:
- Pulse Frequency Range: Optimal 1-120Hz for versatile treatments
- Output Channels: Minimum 6 for professional use
- CE/FDA Certifications: Verify registration numbers
Chapter 3: EMS NEO Quality Control Standards Decoded
Reputable manufacturers comply with:
- ISO 13485 for medical devices
- IEC 60601-1 electrical safety
- 21 CFR Part 803 (FDA reporting)
Chapter 4: Top 10 EMS NEO Supplier Comparison
| Supplier | MOQ | Warranty |
|---|---|---|
| Company A | 5 units | 2 years |
Chapter 5: Procurement Risks & Mitigation Strategies
Common pitfalls include:
- Fake certifications (always verify with issuing bodies)
- Underpowered motors (request lab test reports)
Conclusion: Building Your Supplier Evaluation Framework
Develop a 20-point checklist covering:
- Factory audit results
- Clinical study data
- After-sales support
FAQ Section
What's the MOQ for EMS NEO machines?
Most suppliers require 3-5 units for initial orders.
How to verify supplier certifications?
Cross-check with FDA database or notified bodies.











