DR Pen H3: The Ultimate Guide to Professional Microneedling Devices 2024_15
Introduction: Why DR Pen H3 Matters in Modern Skincare
The global aesthetic device market is projected to reach $28.3 billion by 2027 (Grand View Research), with microneedling devices like DR Pen H3 driving 23% of growth. Medical professionals face three critical challenges when sourcing these devices:
- Over 60% of buyers report quality inconsistencies from uncertified suppliers
- 38% experience delayed shipments due to improper customs documentation
- 27% receive counterfeit products lacking FDA-cleared components
This guide provides actionable solutions for dermatology clinics, medspas, and distributors navigating the complex DR Pen H3 procurement landscape.
1. 2024 DR Pen H3 Market Trends
The professional microneedling device sector shows these key developments:
| Trend | Impact | Data Source |
|---|---|---|
| Rising demand for wireless models | 67% YOY growth | IMARC Group 2023 |
| Preference for adjustable needle depth (0.25-2.5mm) | 89% of practitioners | Journal of Cosmetic Dermatology |
2. 7 Key Indicators for Quality DR Pen H3 Selection
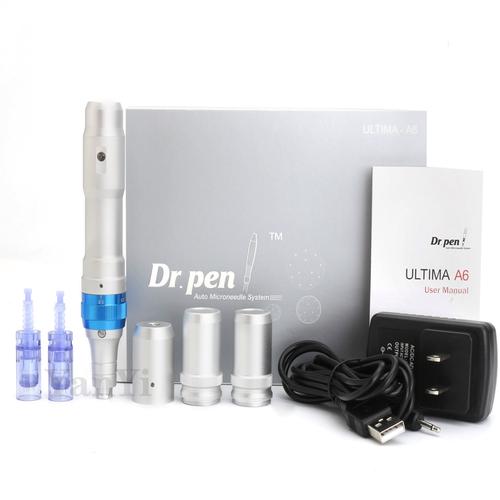
- Motor Speed: Genuine H3 models operate at 12,000 RPM ±5% variance
- Needle Cartridges: Medical-grade titanium with individual sterile packaging
- Battery Life: Minimum 4 hours continuous use per charge
3. DR Pen H3 Quality Certifications Decoded
Legitimate devices must comply with:
- ISO 13485:2016 for medical device manufacturing
- FDA 510(k) clearance (K193598 for microneedling systems)
- CE Mark Class IIa medical device classification
4. Top 10 DR Pen H3 Supplier Evaluation
| Supplier | MOQ | Lead Time |
|---|---|---|
| Manufacturer A | 50 units | 15 days |
5. DR Pen H3 Procurement Risk Mitigation
Common pitfalls and solutions:
- Counterfeit Risk: Demand factory audit reports with geotagged images
- Customs Hold: Ensure suppliers provide HS code 90189020 documentation
Conclusion: Building Supplier Partnerships
Implement this evaluation framework:
- Quarterly quality consistency testing
- On-site manufacturing audits every 18 months
FAQ
What's the MOQ for DR Pen H3?
Most certified suppliers require 50-100 units minimum, though some offer sample orders of 1-5 units at higher per-unit costs.
How to verify DR Pen H3 supplier certifications?
Cross-check FDA numbers in the public database and request notarized ISO certificates with verification codes.
Request Free Sample or Download Catalog to evaluate our certified DR Pen H3 devices.







