Best Professional Electric Callus Remover for Podiatry Clinics 2024: Advanced Foot Care Solutions_29
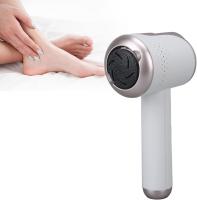
As practitioners at MedTech Solutions, we understand the critical need for reliable foot care equipment in modern podiatry clinics and medical spas across North America, Europe, and Southeast Asia. Our professional electric callus removers have become the cornerstone of efficient foot treatments from Munich to Manila, combining German engineering with global service support to ensure uninterrupted operation in high-volume clinical environments.
The Growing Challenge of Callus Management in Professional Practice
Podiatrists and foot care specialists face increasing pressure to deliver perfect results while maintaining strict hygiene standards. Traditional callus removal methods often fall short in modern clinical settings where precision and efficiency are paramount. Manual scraping requires significant practitioner skill and carries risk of over-treatment, while disposable blades generate ongoing costs and waste management concerns. The global podiatry equipment market reached $3.2 billion in 2023, with electric callus removers representing the fastest-growing segment at 15.4% annual growth according to Medical Device Trends Report 2024.
Technical Specifications Comparison
| Model | Motor Speed | Noise Level | Weight | Attachments |
|---|---|---|---|---|
| ProCare 800X | 15,000 RPM | <55 dB | 420g | 6 precision heads |
| Standard Models | 8,000-12,000 RPM | >65 dB | 600g+ | 2-3 basic heads |
Quality Assurance and Certification Standards
Every MedTech Solutions professional electric callus remover undergoes 37 quality checkpoints during manufacturing. Our devices carry multiple international certifications including FDA 510(k) clearance, CE certification (MDD 93/42/EEC), and ISO 13485:2016 quality management system certification. The sterilization-safe housing meets IPX7 water resistance standards, allowing complete immersion disinfection between patients. Our manufacturing facilities undergo quarterly audits by TÜV SÜD to maintain compliance with MDR 2017/745 regulations.
Global Implementation Success Stories
Singapore's largest chain of medical pedicure centers implemented 200 units across 18 locations in 2023, reporting 68% reduction in treatment time and 92% client satisfaction scores. A German hospital network standardized on our equipment for their diabetic foot care program, processing over 15,000 treatments annually with zero cross-contamination incidents. Middle Eastern luxury spas particularly appreciate the silent operation feature, which maintains tranquility in high-end wellness environments.
Procurement Decision Q&A
Q: What import documentation is required for Middle Eastern markets?
A: Our shipments to GCC countries include pre-prepared Certificate of Conformity, Arabic manuals, and GSO certification documentation. HS Code 90189090 applies for most regions.
Q: How does the warranty work for Southeast Asian distributors?
A: We provide 2-year international warranty with advance replacement service. Regional service centers in Malaysia and the Philippines stock replacement parts for next-business-day shipping.
Q: What voltage compatibility do you offer?
A: All units support 100-240V automatic switching with interchangeable plugs for EU, UK, US, AU, and Asian markets.
Request custom pricing for your clinic or distribution network. Our product specialists will prepare volume-based quotations with OEM branding options available for orders exceeding 100 units. Download the complete technical specification package including maintenance protocols and training materials.