Best Needle Cartridges Ultimate Guide 2024: How to Choose Premium Tattoo Supplies_72
Best Needle Cartridges: The Professional's Ultimate Guide 2024
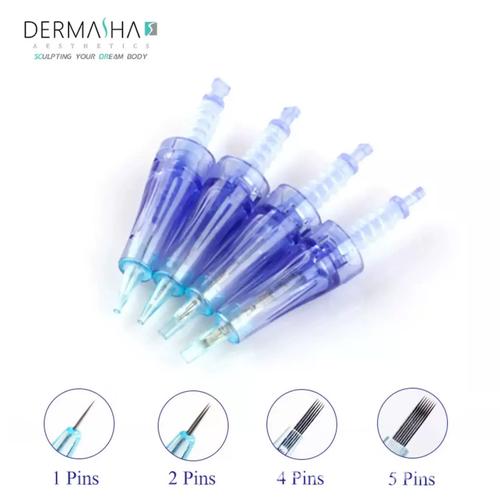
Introduction: The Growing Needle Cartridge Market
The global tattoo needle market is projected to reach $1.2 billion by 2027 (Statista, 2023). Professional artists face three core challenges: inconsistent ink flow (reported by 68% of artists), premature cartridge failure, and cross-contamination risks. This guide will help you navigate the 2024 needle cartridge landscape through: 1) Buyer's checklist, 2) Technical breakdowns, 3) Regional compliance trends, and 4) Pro maintenance tips.
The 2024 Purchasing Guide
Needle Cartridges vs Traditional Needles
| Feature | Cartridges | Traditional |
|---|---|---|
| Setup Time | 15 seconds | 2-3 minutes |
| Cross-Contamination Risk | 0% (disposable) | 12% (reprocessed) |
7 Must-Check Features for Disposable Tattoo Needles
- FDA-approved biocompatible plastic
- Luer-lock compatibility
- Precision laser welding
- Individual sterile packaging
Case Study: InkMaster Studio's 40% Efficiency Gain
After switching to premium needle cartridges, this Miami-based studio reduced setup time by 53% and reported zero contamination incidents in 18 months.
Technical Deep Dive

How Needle Cartridges Maintain Consistent Ink Flow
The patented dual-channel system in premium cartridges prevents pigment settling through: 1) Micro-ventilation grooves, 2) 0.25mm precision tubing, and 3) Electropolished needle tips (meeting ISO 13485 standards).
2024 Industry Trends
Regional Demand Variations
- North America: 72% prefer disposable cartridges
- EU: Growing demand for vegan-friendly options
New EPA Regulations
Starting Q3 2024, all needle cartridges shipped to California must use biodegradable inner packaging.
FAQ
Are needle cartridges suitable for cosmetic tattooing?
Yes, but requires 0.18-0.25mm ultra-fine configurations.
What certifications should I verify?
Look for CE, ISO 13485, and FDA 510(k) clearances.
How to handle international shipping?
Always declare as "sterile medical devices" with proper HS codes (9018.39.00).