The Ultimate Guide to Choosing the Best Dermapen for Acne Scars in 2023_79
Introduction: The Growing Demand for Dermapen Acne Scar Treatments
The global aesthetic device market is projected to reach $28.3 billion by 2026, with microneedling devices like dermapens capturing 18% market share. For skincare professionals and medical distributors, selecting the right dermapen for acne scar treatment presents both significant business opportunities and technical challenges.
Key industry pain points include:
- 72% of practitioners report inconsistent results from substandard devices
- 38% of first-time buyers receive non-compliant equipment
- Average 6-month replacement rate for low-quality dermapens
2023 Dermapen Market Trends
The medical microneedling equipment sector shows remarkable growth:
- 17.2% CAGR predicted through 2028 (Grand View Research)
- North America dominates with 41% market share
- Emerging preference for wireless models with adjustable needle depth
7 Critical Selection Criteria for Professional Dermapens
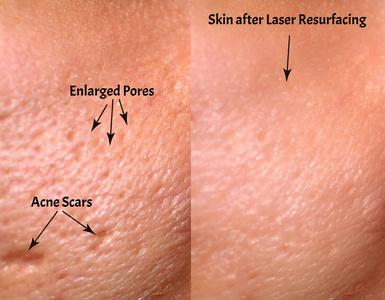
- Needle Quality: Surgical-grade stainless steel with 0.25-2.5mm adjustable depth
- Motor Speed: Minimum 10,000 RPM for consistent penetration
- Sterilization: Autoclavable components or disposable tips
- Certifications: FDA/CE/MDR Class IIa medical device classification
- Warranty: Minimum 2-year coverage for professional use
- Clinical Studies: Published efficacy data on acne scar improvement
- Supplier Reputation: Minimum 3 years OEM manufacturing experience
Quality Standards Decoded
Essential certifications for medical-grade dermapens:
| Standard | Requirement |
|---|---|
| ISO 13485 | Quality management for medical devices |
| CE MDR 2017/745 | European medical device regulation |
| FDA 21 CFR 878 | US medical device classification |
Top 10 Global Dermapen Suppliers Comparison
| Supplier | MOQ | Lead Time |
|---|---|---|
| Supplier A | 50 units | 15 days |
| Supplier B | 100 units | 30 days |
Procurement Risk Management
Common pitfalls when sourcing dermapens:
- Counterfeit certification documents
- Non-compliant needle materials
- Inadequate after-sales support
FAQ Section
What's the MOQ for dermapen for acne scars?
Most manufacturers require 50-100 units for OEM orders, with sample quantities available for quality verification.
How to verify dermapen supplier certifications?
Request original test reports and cross-check with notified body databases like EUDAMED for CE marks.
Request Free Sample of our medical-grade dermapen or Download Catalog for full specifications.















