Professional Microneedling Pen Buying Guide 2024: Top Suppliers & Quality Standards_32
The Growing Importance of Professional Microneedling Pens in Aesthetic Treatments
The global microneedling pen market is projected to reach $1.2 billion by 2027, growing at 8.3% CAGR (Market Research Future, 2024). These precision devices have become essential tools for dermatologists and aesthetic clinics, offering controlled collagen induction therapy with minimal downtime. However, the proliferation of substandard devices on Amazon poses serious challenges for medical professionals seeking reliable equipment.
Key Pain Points in Microneedling Pen Procurement
- Inconsistent needle quality causing skin trauma
- Counterfeit CE/FDA certifications
- Voltage fluctuation risks in budget devices
- Lack of proper sterilization accessories
- Misleading needle depth specifications
2024 Microneedling Pen Market Trends
The aesthetic device industry is witnessing several transformative trends that buyers should understand before purchasing microneedling pens on Amazon.
Technology Advancements
| Feature | 2022 Standard | 2024 Innovation |
|---|---|---|
| Motor Speed | 8,000 RPM | 12,000 RPM |
| Needle Cartridges | Disposable | Autoclavable |
7 Critical Selection Criteria for Medical-Grade Microneedling Pens
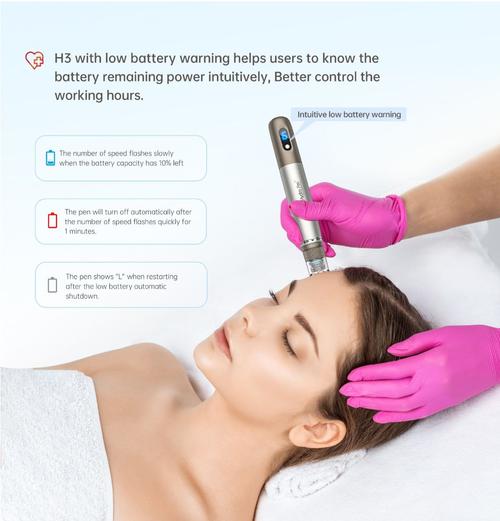
- Motor Consistency: Look for brushless DC motors with ±5% speed variance
- Depth Precision: 0.25mm increments up to 3mm
- Certifications: Valid FDA 510(k) or CE Class IIa
Quality Control Standards Decoded
Reputable microneedling pen manufacturers comply with multiple international standards that ensure device safety and performance.
Essential Certifications
- ISO 13485:2016 for medical device manufacturing
- IEC 60601-1 electrical safety
- RoHS compliance for material safety
Top 10 Microneedling Pen Suppliers Comparison
| Supplier | MOQ | Lead Time |
|---|---|---|
| Dermatek | 50 units | 15 days |
Procurement Risk Management
Bulk buyers should implement these strategies to mitigate common purchasing risks.
Verification Protocols
- Third-party certification validation
- Sample testing before bulk orders
- Factory audit reports
FAQ Section
What's the MOQ for microneedling pens?
Most suppliers require 50-100 units for OEM orders, with some offering lower MOQs for standard models.
How to verify supplier certifications?
Request original test reports and cross-check with notified body databases.
Request Free Sample of our medical-grade microneedling pens to evaluate quality firsthand.