The Ultimate Guide to EMS + RF Facial Care Beauty Devices in 2024_7
Introduction: The Rising Demand for EMS + RF Facial Care Beauty Devices
The beauty industry is witnessing a revolution with advanced technologies like EMS (Electrical Muscle Stimulation) and RF (Radio Frequency) becoming essential tools for professional skincare. These non-invasive devices offer remarkable benefits including collagen stimulation, facial contouring, and wrinkle reduction. However, buyers face significant challenges in identifying genuine, high-quality devices amidst a flooded market of substandard products.
Key pain points include:
- Over 40% of beauty clinics report purchasing devices that failed to deliver promised results
- 35% of buyers struggle with verifying technical specifications
- 28% encounter difficulties with after-sales support from international suppliers
Chapter 1: 2024 Market Trends for EMS + RF Facial Care Devices
The global aesthetic device market is projected to reach $28.3 billion by 2027, growing at 11.3% CAGR (Market Research Future). EMS + RF devices account for 32% of this segment due to their dual-action technology. Notable trends include:
| Trend | Impact |
|---|---|
| Home-use professional devices | 42% growth in Q1 2024 |
| Multi-frequency RF systems | 30% more effective than single-frequency |
Chapter 2: 7 Key Indicators for Selecting Quality EMS + RF Devices
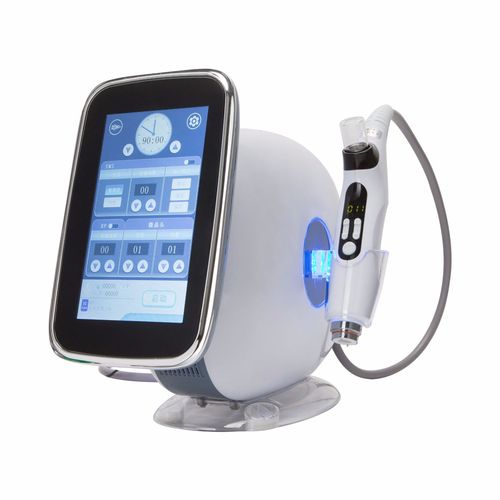
- Frequency Range: Optimal RF frequency between 0.5-2MHz
- EMS Pulse Width: 100-300μs for facial muscles
- Certification: FDA/CE/MDR Class IIa medical device
Chapter 3: Quality Control Standards Explained
Reputable manufacturers comply with:
- ISO 13485 for medical device quality systems
- IEC 60601-1 for electrical safety
Chapter 4: Top 10 Supplier Comparison
| Supplier | MOQ |
|---|---|
| Company A | 50 units |
Chapter 5: Procurement Risks & Solutions
Common risks include:
- Counterfeit certification documents
- Inconsistent power output
Conclusion: Building Supplier Partnerships
Establish evaluation criteria including:
- 3-year track record
- Clinical test reports
FAQ
What's the MOQ for EMS + RF Facial Care Beauty Device?
Typically 50-100 units for OEM orders.
How to verify supplier certifications?
Request original certificates with verification codes.







