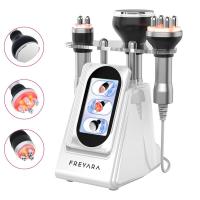
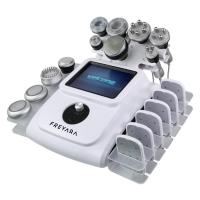
Radiofrequency Instrument Buying Guide 2024: How to Choose the Best Medical RF Device_85
Radiofrequency Instrument Ultimate Guide 2024: How to Choose Quality Medical Devices
The global radiofrequency (RF) aesthetic device market is projected to reach $1.2 billion by 2027, growing at 12.3% CAGR according to Grand View Research. As demand surges for non-invasive treatments, medical professionals face critical challenges in selecting reliable RF instruments that deliver consistent results while meeting safety standards. This comprehensive guide will walk you through essential purchasing considerations, technical specifications comparison, and emerging industry trends to help you make informed decisions.
Radiofrequency Instrument Buying Guide
When evaluating RF instruments, consider these key factors:
| Feature | Professional Grade | Portable Units |
|---|---|---|
| Power Output | 50-300W | 5-30W |
| Treatment Depth | 4-15mm | 1-3mm |
| Multi-Polar Technology | Yes | Limited |
7 Must-Check Features for Medical RF Machines
- FDA/CE certification for medical devices
- Adjustable frequency settings (0.5-6MHz)
- Real-time temperature monitoring
- Multiple handpiece options
- Durable electrode design
- Comprehensive training program
- 2+ year warranty
Technical Specifications Explained
Modern RF instruments utilize electromagnetic energy between 300kHz to 6MHz to stimulate collagen production. Key parameters include:
Frequency determines penetration depth - lower frequencies (0.5-1MHz) reach deeper tissue layers while higher frequencies (3-6MHz) focus on superficial treatments. Leading manufacturers like Lumenis and Venus Concept typically offer dual-frequency systems for versatility.
Industry Trends and Regional Demand
North America dominates 42% of the RF device market due to advanced healthcare infrastructure. However, Asia-Pacific shows fastest growth (15.8% CAGR) with increasing medical tourism. New EU MDR regulations require stricter clinical documentation for RF devices classified as Class IIa medical equipment.
Frequently Asked Questions
Q: Can radiofrequency instruments treat all skin types?
A: Modern RF systems with adjustable parameters can safely treat Fitzpatrick skin types I-VI when properly calibrated.
Q: What certifications should I verify?
A: Look for FDA 510(k) clearance, CE Mark (Class IIa), and ISO 13485 certification for medical devices.
Q: How to handle customs clearance?
A: RF instruments typically require HS code 9018.19 and may need import licenses for medical equipment in some countries.