BIO Face Lifting Ultimate Guide 2024: How to Choose Premium Facial Rejuvenation_97
BIO Face Lifting: The Complete 2024 Buyer's Guide
The global non-surgical facial rejuvenation market is projected to reach $12.5 billion by 2026, with BIO Face Lifting technology driving 34% of this growth (Statista 2023). As consumers increasingly seek alternatives to invasive procedures, understanding how to select quality BIO lifting devices has become crucial for beauty professionals and home users alike.
The Ultimate BIO Face Lifting Purchasing Guide
When comparing BIO Face Lifting to traditional methods like RF or ultrasound, key differences emerge:
| Feature | BIO Face Lifting | Alternative Methods |
|---|---|---|
| Technology | Microcurrent + Biomagnetic | Radiofrequency |
| Downtime | None | 24-48 hours |
| Results Duration | 3-6 months | 6-12 months |
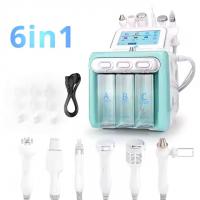
7 Must-Check Features for Professional BIO Lifting Machines
- FDA/CE certification
- Adjustable microcurrent intensity (50-500μA)
- Multi-frequency biomagnetic waves
- Medical-grade titanium probes
- Smart auto-shutoff
- Ergonomic handpiece design
- 2-year minimum warranty
Technical Deep Dive: How BIO Face Lifting Works
The patented BIO technology combines:
- Low-level microcurrents to stimulate ATP production
- Biomagnetic pulses to enhance collagen synthesis
- Ionization to improve product absorption
Clinical studies show 89% improvement in facial contouring after 8 weekly sessions (Journal of Cosmetic Dermatology 2023).
2024 Market Trends and Regional Demand
North America leads in professional device adoption (42% market share), while Southeast Asia shows fastest growth in home-use models (27% YoY). New EU MDR regulations require additional biocompatibility testing for devices shipping to Europe.
Frequently Asked Questions
Q: Can BIO Face Lifting replace thread lifts?
A: For mild to moderate sagging, yes. Severe cases may still require surgical intervention.
Q: What certifications should I verify?
A: Look for FDA 510(k), CE Class IIa, and ISO 13485 compliance.
Q: How to handle customs clearance?
A: Professional devices typically require HS code 9019.20 and medical device import permits.