Beauty Equipment Ultimate Guide 2024: How to Choose Professional-Grade Tools_64
The Complete Guide to Professional Beauty Equipment in 2024
The global beauty equipment market is projected to reach $135 billion by 2027, growing at 13.2% CAGR according to Grand View Research. As salon owners and skincare professionals face increasing pressure to deliver clinical-grade results, selecting the right beauty equipment becomes critical. This comprehensive guide will walk you through equipment selection, technical specifications, industry trends, and practical FAQs to help you make informed purchasing decisions.
Beauty Equipment Buying Guide: 7 Must-Check Features
When investing in professional beauty equipment, these are the essential factors to consider:
| Feature | Entry-Level | Professional-Grade |
|---|---|---|
| Motor Power | 0.5-1HP | 1.5-3HP |
| Warranty | 1 year | 3-5 years |
Case Study: How Spa XYZ Increased Revenue 40% With Advanced Microcurrent Devices
By upgrading to medical-grade microcurrent equipment, this Miami spa reduced treatment time while achieving better facial contouring results. Their $15,000 investment paid off within 8 months through premium service pricing.
Technical Deep Dive: Understanding Beauty Equipment Specifications
Modern beauty devices incorporate several advanced technologies:
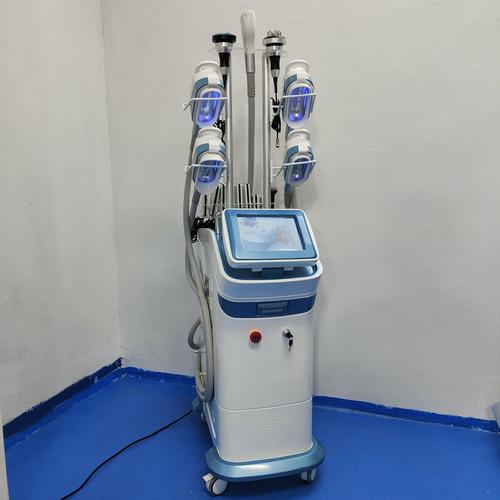
RF (Radio Frequency) systems typically operate at 0.3-10MHz, with higher frequencies providing more superficial heating for collagen stimulation. Look for equipment with adjustable frequency settings for customized treatments.
2024 Beauty Equipment Trends: What Professionals Need to Know
The European market shows strongest demand for multifunctional devices (78% growth YoY), while North America leads in microcurrent technology adoption. New EPA regulations will require all electrical beauty equipment to meet Energy Star standards by Q3 2024.
Beauty Equipment FAQ
Q: Can I use salon equipment for mobile services?
A: Many jurisdictions require special certifications for portable equipment. Check your local regulations regarding electrical safety standards for mobile operations.
Q: What certifications should I look for?
A: FDA clearance (for US market), CE marking (Europe), and ISO 13485 certification indicate medical-grade quality for devices making therapeutic claims.













-200x200.jpg)